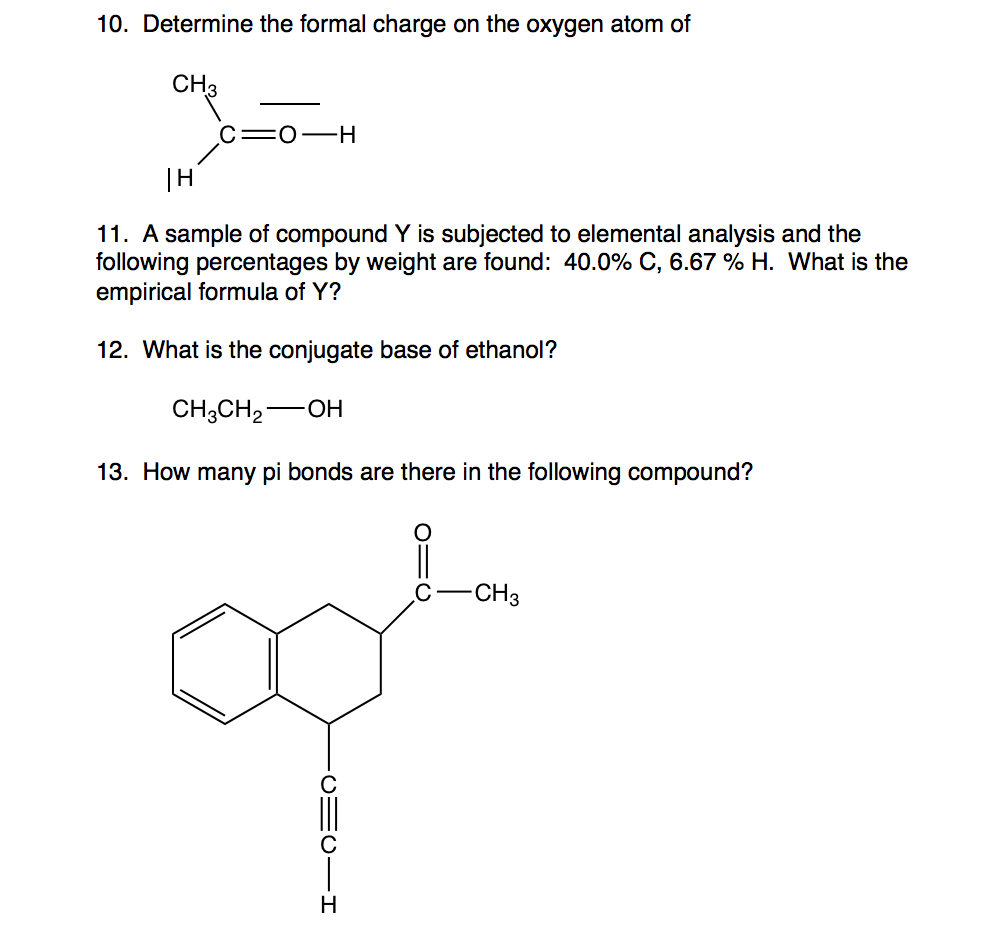
Fluorine is extremely reactive and reacts directly with all elements except helium (He), neon (Ne) and argon (Ar). Its oxidation state is always -1 except in its elemental, diatomic state (in which its oxidation state is zero). Fluorine also has a relatively small atomic radius. 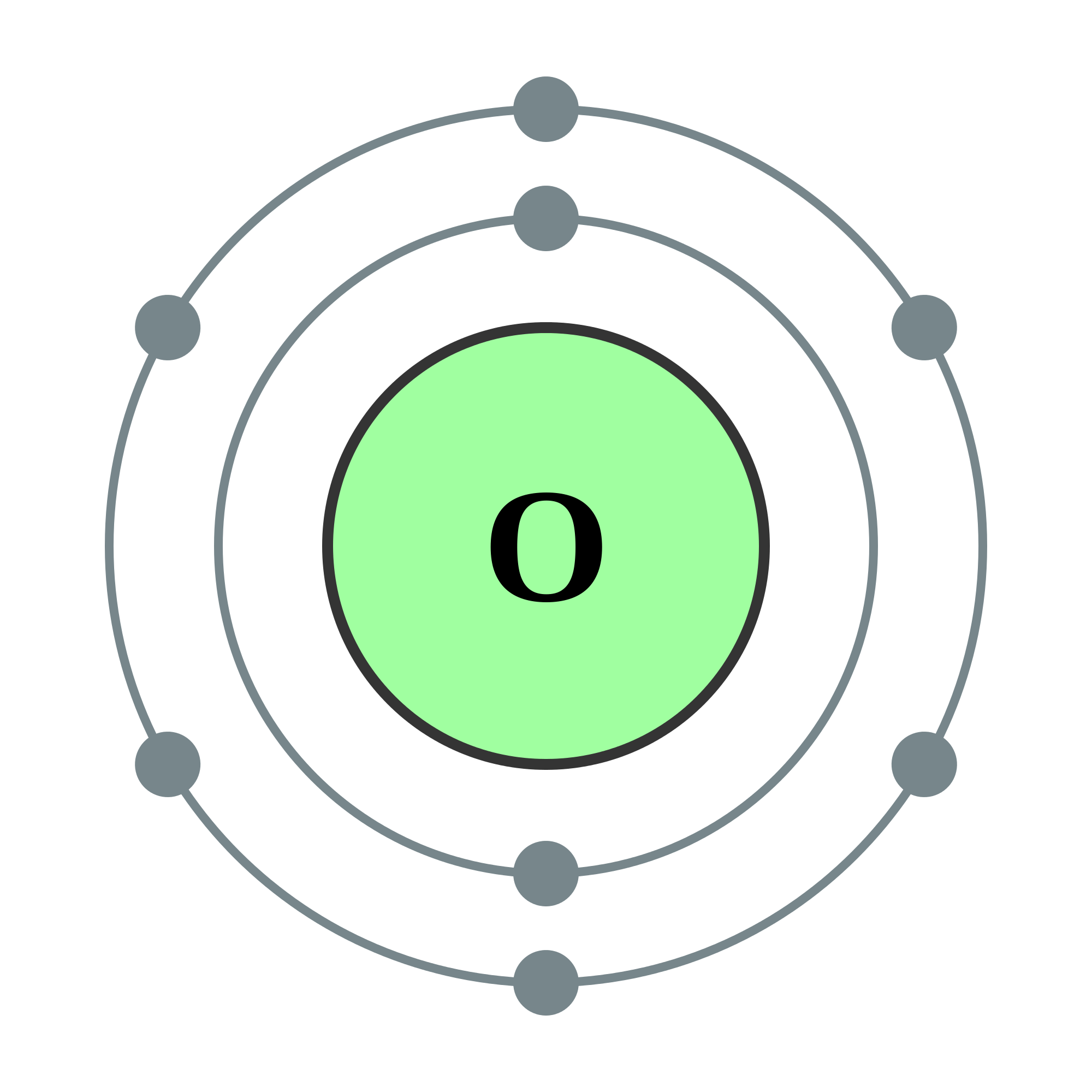
It appears as a pale yellow gas at room temperature. Fluorine is the most electronegative element in the periodic table. Fluorine exists as a diatomic molecule in its free state (F 2) and is the most abundant halogen found in the Earth's crust. Elemental fluorine was first discovered in 1886 by isolating it from hydrofluoric acid. Lastly, the halogens are also relevant in daily life, whether it be the fluoride that goes into toothpaste, the chlorine that disinfects drinking water, or the iodine that facilitates the production of thyroid hormones in one's body.įluorine - Fluorine has an atomic number of 9 and is denoted by the symbol F. Halite is the mineral name for rock salt, a natural mineral consisting essentially of sodium chloride (NaCl). Together these words combine to mean "salt former", referencing the fact that halogens form salts when they react with metals. The name "halogen" is derived from the Greek roots hal- ("salt") and -gen ("to form"). Halogen elements can cross-link to form diatomic molecules with polar covalent single bonds.Ĭhlorine (Cl 2) was the first halogen to be discovered in 1774, followed by iodine (I 2), bromine (Br 2), fluorine (F 2), and astatine (At, discovered last in 1940). When a halogen atom is substituted for a covalently-bonded hydrogen atom in an organic compound, the prefix halo- can be used in a general sense, or the prefixes fluoro-, chloro-, bromo-, or iodo- can be used for specific halogen substitutions. When in the -1 oxidation state, with carbon or nitrogen in organic compounds, the halogens often form single bonds. Therefore, most of the chemical reactions that involve halogens are oxidation-reduction reactions in aqueous solution. 
Replacing the -ine ending with an -ide ending indicates the presence of halide anions for example, Cl - is named "chloride." In addition, halogens act as oxidizing agents-they exhibit the property to oxidize metals. In these compounds, halogens are present as halide anions with charge of -1 (e.g., Cl -, Br -, etc.). All halogens form Group 1 salts with similar properties. As a general rule, fluorine is the most reactive halogen and astatine is the least reactive. However, halogens readily combine with most elements and are never seen uncombined in nature. The bonds in these diatomic molecules are non-polar covalent single bonds. Halogens form diatomic molecules (of the form X 2, where X denotes a halogen atom) in their elemental states.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
